Dr. Shore Q&A by Tinnitus Hub
Support
Tinnitus Hub

I'm Hazel, Tinnitus Hub's Director. I got tinnitus in 2017 and have volunteered for Tinnitus Hub ever since. We bring value to the tinnitus community for example with our:
We are volunteer-run with no salary costs. But to keep doing what we do, we must cover our expenses, and this is where you can help today.
As a registered 501(c)(3) nonprofit, donations in the US are tax deductible.
Every contribution to our cause, regardless of its size, holds immense value. It's a testament to your belief in us, your faith in our ability to make a change.
Thank you!
Dr. Susan Shore Q&A
Tinnitus Hub is proud to present a Q&A with Dr. Shore, a neuroscientist who has been working on her bimodal stimulation treatment for tinnitus for over a decade.
On our Tinnitus Talk community, the thread discussing her research has seen over 1 million views. Now, Dr. Shore finally answers many of your burning questions!
We received nearly 900 questions from you, which we worked hard to condense and categorize. We hope you will get the answers you are looking for.
Please note that Tinnitus Hub (operator of Tinnitus Talk) does not endorse or promote Dr. Shore's device, nor do we have any commercial ties with her or Auricle Inc. We do not have the expertise nor time (since we are run by volunteers who suffer from tinnitus) to provide an in-depth assessment of the pros and cons of the treatment. We encourage everyone to educate themselves on risks and benefits before trying any tinnitus treatment. We have invested our time and effort in presenting this Q&A as a service to the tinnitus community, in response to the overwhelming interest in Dr. Shore's research.
Kindly refrain from contacting Dr. Shore directly, so she can focus on her work to get the device to market as quickly as possible.

Answers by
Dr. Susan Shore
Chief Scientific Officer, Auricle Inc.
Professor Emerita, University of Michigan.
General Background
What is bimodal stimulation? How does the device work?
Bimodal stimulation means providing two different modes of stimulation. Various groups of researchers are working on different bimodal stimulation treatments for tinnitus. One of these groups is Dr. Susan Shore and her team at the University of Michigan. Auricle, Inc. is the licensee of the intellectual property developed by Dr. Shore’s Lab.
The device that Dr. Shore created provides sound stimulation to the ear combined with mild electrical stimulation of the neck or jaw. This treatment is based on many years of basic science studies that demonstrate that tinnitus originates in the first brain region that connects to the ear, the cochlear nucleus. The cochlear nucleus is where sound signals and somatosensory (touch) signals come together. The precisely timed auditory-somatosensory stimulation provided by Shore’s treatment has been shown to significantly reduce the spontaneous firing rates of neurons that carry the tinnitus signals to higher regions of the brain, thus inhibiting the perception of tinnitus in those higher brain regions.
The take-home treatment device was used for 30 minutes per day for 6 weeks in the latest human trial. Each device provided a bimodal stimulation treatment in which the auditory stimulus was matched to the patient’s tinnitus sound and the electrical stimulation was adjusted to the threshold of sensation. You can read Dr. Shore’s latest published paper to understand more. The paper provides the full results of Dr. Shore’s clinical trial in which her team tested the treatment on 99 participants. The results showed statistically significant reductions in tinnitus loudness and impact on life.
Regulatory Approval
Has the device yet been submitted to the FDA for regulatory approval? Yes or no? If you can't divulge this information, why not? Has Auricle Inc. received confirmation from the FDA that the design of the latest trial was adequate to support an application for regulatory approval of the device, or is there still a chance the agency may deem the results unsatisfactory and request yet another trial?
I am not in a position to discuss or otherwise confirm or deny anything concerning Auricle and the FDA. The company is certainly working on a regulatory clearance, but it is not making public comments concerning the process. The company has provided the following general statement.
Auricle, Inc., the privately-held company that is the exclusive licensee of the patents related to The Shore Lab’s tinnitus therapy, is engaging with the FDA concerning a regulatory clearance. The Company is not, however, providing guidance or other information concerning the status of its engagement. It is not appropriate for companies in the midst of negotiations with the FDA to forecast or otherwise publicly speculate on the terms and conditions that may lead to a clearance—this can cause potential issues with the agency as well as set potentially inappropriate expectations with other stakeholders and interested parties. In this same vein, the company is not providing commentary about agency guidance and the specific terms and conditions that would lead to a clearance.
The Polish Tinnitus Association would like to offer Auricle Inc. their help with registration and commercialization of the device in Poland. Are you interested in co-operation?
Auricle thanks the Polish Tinnitus Association for their interest and desire to provide assistance, but at this time the Company is focused on an FDA clearance. Europe is an important market for the Company, but the European Union’s updated Medical Device Regulations reveal that the pathway to clearance is neither simple nor fast relative to the United States. For certain, the Company will focus resources on an EU clearance in the future.
Availability & Pricing
If FDA approval is successful, does Auricle Inc. already have the financing, business infrastructure, and manufacturing capacity in place to bring the device to market?
As a privately held company, Auricle is raising the resources based on its business plan and milestone objectives. It is not providing public guidance on questions pertaining to commercialization.
Has the federal government or any other other governmental institution taken interest in the device? Considering tinnitus is the #1 veterans' disability, has the government offered more funding to you?
Outside of the engagement with the FDA, there are no on-going collaborations with any other governmental division or entity.
When will the Auricle Inc. website go live?
Auricle has no immediate plans for a website.
How would people be able to access the device? Will it need to be set up by an audiologist or other medical professional? Is a one-off set up enough or do you need regular appointments throughout the treatment? Can further adjustments be made at home by the patient?
The device needs to be fitted for each patient by a trained healthcare professional (for example, an audiologist). Patients are expected to have some level of follow-up to monitor results, and there are no adjustments made by the patient directly.
Do you plan to launch the device first in the US? What geographic locations would follow next? After US market launch, can patients fly to the US to get access to the treatment? How long would they need to stay in the US, and could they bring the device to their home country afterwards?
Auricle is not providing commentary concerning the sequencing of global markets.
How much will the device cost? Can you estimate a rough price range in $USD?
Auricle is not providing commentary concerning pricing, but it is the company’s hope to make the therapy available to as many tinnitus sufferers as possible at pricing that is accessible to most.
Do you think insurance plans will cover the device? If not, will there be financing plans?
Whether or not insurance providers offer therapy coverage will be dependent on the individual provider. In the US, because there is no established standard of care for tinnitus treatment and therefore no CPT (‘Current Procedural Therapy’) codes for reimbursement, it will require a multi-year effort for providers to offer reimbursement. There are many established and possible financing models, however, that should contribute to making the therapy accessible to most.
Compassionate Use
Would you consider a pathway of compassionate care use purchase if FDA submission is being delayed? In other words, could a patient, with comprehensive doctor/audiologist documentation of level of debilitation and need for treatment, purchase the device after signing of said documents confirming understanding it is pending FDA approval, and gain access to education for device use?
The terms and conditions for ‘compassionate care use’ per the FDA are extremely specific, and it is only granted for immediately life-threatening diseases where there are no cleared therapies. There is now at least one FDA-cleared device to treat the symptoms of tinnitus, so compassionate care use of other non-FDA cleared approaches is challenging. In addition, Auricle does not have manufactured devices and the requisite infrastructure to support device use outside of investigational research.
Tinnitus Mechanisms/Models
Why can some people modulate their tinnitus and others not?
A majority of people with tinnitus can modulate. These modulations reflect the connections between the somatosensory and auditory systems. In those who cannot modulate, we assume those connections are still there but perhaps not as strong. The preclinical (animal model) studies indicate that projections from somatosensory centers to the cochlear nucleus are present in 100% of animals tested.
Given some theories on the relationship between tinnitus and visual snow syndrome, could your treatment theoretically help visual snow syndrome as well?
It is not possible to make this connection as the basic science studies were done on the auditory system and not the visual system. The circuitry of the two sensory systems is different so a new set of studies would need to be done to investigate visual snow and a comparable visual based treatment.
What are your thoughts on “residual inhibition”? And how relevant, if at all, do you see it being to the device’s effectiveness, i.e. does experiencing residual inhibition possibly influence how effective the device will be? (Not everyone with tinnitus experiences this phenomenon.)
Residual inhibition is a real phenomenon that occurs in many subjects. We did not specifically study the effect of residual inhibition on auditory-somatosensory integration. It is unlikely that residual inhibition plays a role in our bisensory treatment because the long term suppression that we see with specific auditory-somatosensory orders and intervals is based on long term plasticity in fusiform cells of the cochlear nucleus, a process that is independent of residual inhibition.
Do you believe that all forms of subjective tinnitus (excluding those caused by mechanical issues) are generated in the dorsal cochlear nucleus (DCN)?
All signals must be processed by the cochlear nucleus (including the dorsal cochlear nucleus) before ascending to the rest of the auditory system. Thus, the tinnitus signal that fusiform cells produce is passed along to the rest of the brain. So yes, the tinnitus signal originates in the cochlear nucleus. More central centers may modify the signal that they receive from the cochlear nucleus, including descending pathways.
How did you know in the animal studies that the guinea pigs had somatic tinnitus?
We don’t know that the guinea pigs had somatic tinnitus per se. We do know that somatosensory stimulation could modify auditory signals in all of the guinea pigs. Thus, we chose those who could modulate their tinnitus as our first test group as this was the group showing evidence of somatosensory innervation to the auditory system.
Treatment/Study Specific Questions
Eligibility and/or Efficacy for Different Patient Groups
Does time since tinnitus onset matter? Will the treatment be equally effective for someone who has had tinnitus for weeks/months versus someone who has had tinnitus for decades?
Our studies have shown no influence of tinnitus duration on treatment efficacy.
Does the patient's age matter? Could it be that the treatment works better in a younger person?
The age of the patient per se did not influence the outcome of the studies.
Do you need to be able to match your tinnitus tone to use the device? What if you don't have a stable tinnitus tone, but e.g., electrical noises, crickets, screeching, or multiple tinnitus tones in various frequencies? Can all these different tones and/or sounds be treated at the same time? Does the treatment work better for a certain kind of tinnitus, such as tonal high frequency?
The TinnTester interactive tinnitus matching software program is designed to match different kinds of tinnitus and has shown high test-retest reliability. Most tinnitus is narrow band or wide band, meaning it is comprised of multiple frequencies – called the tinnitus spectrum. We matched the tinnitus spectrum and presented this as the sound part of the bisensory stimulus. We did not see differences based on spectrum.
Could the treatment be effective for typewriter tinnitus?
We did not assess typewriter tinnitus – this would need to be assessed In the future.
Could the treatment help with pulsatile tinnitus?
We did not assess pulsatile tinnitus – this was an exclusion criterion as pulsatile tinnitus can be linked to cardiovascular system, so may have a different etiology to other types of tinnitus.
Did any of the trial participants have loudness or pain hyperacusis? Do you think the treatment is appropriate for patients with tinnitus who also present with either type of hyperacusis? Could the treatment improve the hyperacusis as well?
The majority of patients did not report hyperacusis.
If a patient has reactive tinnitus that gets worse in response to external stimuli, are they not an ideal candidate?
We did not assess this type of tinnitus.
What if you have bilateral severe to profound hearing loss in the same frequency as your tinnitus? Will it impact the treatment?
At present, bilateral severe to profound hearing loss would preclude use of the device as the signal needs to be heard to be effective. The study only included up to moderate hearing loss.
What if you have unilateral severe to profound hearing loss along with unilateral tinnitus in your left ear, could you take advantage of your right ear with no hearing loss and tinnitus, and still expect results?
Yes as long as the sound can be heard in one ear this can likely still be effective.
Are hearing aids a contra-indication for the treatment?
We excluded subjects who had used a hearing aid in the 6 months preceding trial enrollment so that any potential treatment impact would be eliminated. But there is no reason why hearing aid use would be a contra-indication provided that the patient’s hearing loss threshold is within our prescribed range.
Do you know if a tinnitus patient with a cochlear implant might benefit from the device?
We do not know this at this time. Research would have to explore this paradigm.
Can the treatment help if the patient cannot modulate their tinnitus by jaw/neck/head/other movements? If not, are there plans for a treatment that helps these patients?
It is possible that the treatment could help those who cannot modulate because the preclinical (animal) studies show that after cochlear damage, all animals show somatosensory reinnervation of the cochlear nucleus. We have not yet tested those subjects.
Does the cause of the tinnitus matter in terms of expected treatment efficacy (noise-induced, age-related, ototoxicity, Menière's, otosclerosis, head trauma, etc.)?
Our inclusion criteria for our studies only specified constant, bothersome tinnitus, not what caused the tinnitus. Since the most frequent factor associated with tinnitus is hearing loss due to noise over exposure (including hidden hearing loss), many of our patients had noise induced hearing loss (or hidden hearing loss). Meniere’s disease was excluded from our study because tinnitus in Meniere’s disease patients is intermittent.
Should the original cause of the tinnitus (e.g., TMJD, neck tension) be resolved beforehand to improve the therapeutical results of the device?
It is likely that even patients with somatosensory involvement in their tinnitus also have some degree of cochlear damage, so resolving the TMD may not be necessary for the treatment to work.
Safety
What kind of side effects did the trial participants encounter? What were the most severe ones?
One person had minor redness on their skin due to repeated tape use without sufficient electrode gel. This went away when instructions were followed.
What are the contraindications (medications, conditions) for using the device?
The device is intended for tinnitus patients that are able to modulate their tinnitus with a somatic maneuver. They also must be able to hear the auditory stimulus, so persons with mild to moderate hearing loss will generally be fine (severe hearing loss patients may be contraindicated). We excluded patients in the trial who had Meniere’s Disease, acoustic neuromas, and some other specific conditions. Patients with these conditions may not be suitable for our device.
Were the reported cases where people’s tinnitus worsened long lasting or even permanent? Or did their tinnitus return to baseline?
Only one patient experienced tinnitus worsening possibly associated with using the device during active treatment. Other instances of worsening were related to noise overexposure events during the trial (eg. attending a rock concert).
These returned to within one std. deviation of baseline.
Will the device be safe to use in tinnitus patients with metal implants placed in their head/neck (e.g. venous sinus stents and coils, arterial stents/coils, jugular vein stents, cochlear implants, ventriculoperitoneal shunts, and brain implants)?
It will probably be ok but safety testing will have to be done for these specific cases.
Some tinnitus patients are worried about using headphones. Could the audio stimulation part of the treatment negatively impact your hearing?
No, our device has hardware and software limits to prevent over stimulation.
Treatment Mechanisms/Protocols
Why is the treatment's sound applied only to one ear, and not both?
The reason for this was that we derived the treatment from the preclinical studies in which we damaged only one ear and then treated that side. But we found in the humans that treating one side reduced tinnitus perceived in both ears. The reason for this is because there are neural pathways between the two cochlear nuclei, where the tinnitus is generated and where the treatment site of action occurs.
Would it be possible for those with both severe hyperacusis and severe tinnitus to use in-ear hearing protection and the device simultaneously; which is to say, can we hold the device close to our ears without inserting the in-ear component of the device?
The treatment should only be used as prescribed, with the earphone inserted. However, the level of the sound can be adjusted to be comfortable for each person, as was done in the trials.
Do the treatment sessions require absolute focus on the stimulation, or can you work and be engaged in other matters at the same time?
The subjects were told to sit in a quiet room while they were using the device (only 30 min per day). They were not told to concentrate on the treatment.
Why is the time between the sound and electrical stimuli so important? How important is the sound versus the electrical stimulation? Would either one of them help if they were used independently from one another or does the success for the device depend entirely on the use of both together?
The sound and the electrical stimulation are equally important. It is in fact the combination of the sound with the electrical stimulation in a precise order and time interval that produces the long-term depression that reduces the tinnitus. The precise timing was derived from the preclinical studies in which we recorded from fusiform cells to discover which timing and order would produced the most long term depression.
In theory, could the treatment be more effective when used for longer periods of time than in the study or when re-applied periodically? Will the suggested usage time be 6 weeks?
It is possible that longer times than 6 weeks may be even more effective.
Study Design/Methods
Why did you use a crossover study design?
A crossover study is the strongest possible design for a clinical trial, but only if there is recovery (or return to baseline) after the first treatment period during the washout phase. This worked well for the first pilot study with 4 weeks of treatment (Marks et al, 2018). For that study, the majority of subjects’ tinnitus had mostly returned to baseline after the 4 week washout. For the second study we used 6 weeks of treatment and 6 weeks of washout before the crossover as we expected that we would see a return to baseline after 6 weeks of washout. We did not expect that the treatment would work so well after 6 weeks that there was no return to baseline during the washout. What we saw was that the treatment after 6 weeks was so long lasting that we did not see recovery. In fact, we saw a continued treatment effect up to 12 weeks (and beyond). For that reason the crossover did not work for the second study. This is called a “crossover effect” and it means that the second treatment period was biased by the first period. When that happens, it is not statistically valid to analyze the second half of the study. That is why we focused our analysis only on the first period.
Could you clarify what exactly is “TinnTester” and has it been third party validated? And what method of testing is it utilizing to verify perceived tinnitus noise levels? There seems to be 2 different methodologies of tinnitus volume level testing (e.g., a more basic method using real world sound levels measured like any other sound and a more tinnitus specific one that appears to be a measure of the actual raw volume of the tinnitus itself that uses your hearing loss as a basis).
TinnTester is a software suite designed by Larry Roberts to measure psychophysical attributes of tinnitus including loudness and likeness at several frequencies. The TinnTester reports loudness measurements in dB SPL which can also be expressed as sensation level (SL) by comparing to the audiogram. There have been a number of studies showing test-retest reliability.
The paper states "In the pilot and the present studies, all participants reported quickly habituating to the electrical stimulus, which was close to threshold, making the stimulus difficult to detect. Participants stated that they could not tell whether they were receiving the control or active treatment". This statement appears contradictory, i.e. how can you habituate to the stimulus if you can't detect it, and anecdotal. Is there a more scientific way of assessing the degree to which participants could detect the stimulus?
Prior to using the device subjects were tested to find out their electrical stimulation thresholds. The stimulation level was then set to just above the threshold, which was barely detectible or perceptible. We didn’t have an objective test for detection. We interpreted subject statements that they could not tell which treatment they were getting to be evidence that they had habituated to (or become accustomed to) the electrical stimulus. Keep in mind that the electrodes were in place for the active and sham treatments and the tape itself produced some sensation, likely helping with the blinding.
Outcome Measures and Analysis of Results
The minimal clinically significant change score for Tinnitus Handicap Index is 7 points, as proposed by Zeman et al. Why did your study use 5 points?
In reviewing the literature, we found many papers using different thresholds of meaningful change for the THI.
We utilized Adamchic et al, 2012, who provided their calculation methodology:
https://link.springer.com/article/10.1186/1477-7525-10-79
When determining the efficacy of a treatment, the paper compares the TFI, THI, loudness statistics with the baseline rather than the control. Wouldn't it make more sense to compare to the control? Maybe that wasn't statistically significant?
It is important to compare a subject’s tinnitus with their own baseline as we want to see if their tinnitus got better. We compared both the active and sham treatments with the subject’s baseline at the beginning of the treatment.
What were the mean changes in THI at 6 and 12 weeks from baseline for the active and control arms? As a primary endpoint, how come the THI data is not shown for Supp. Fig 4.? Can that be provided?
We were limited in the number of figures that we could include. Mean change for THI at 6 weeks (PP) is -9 for active -2 for sham. For ITT -7 for active and -2 for sham.
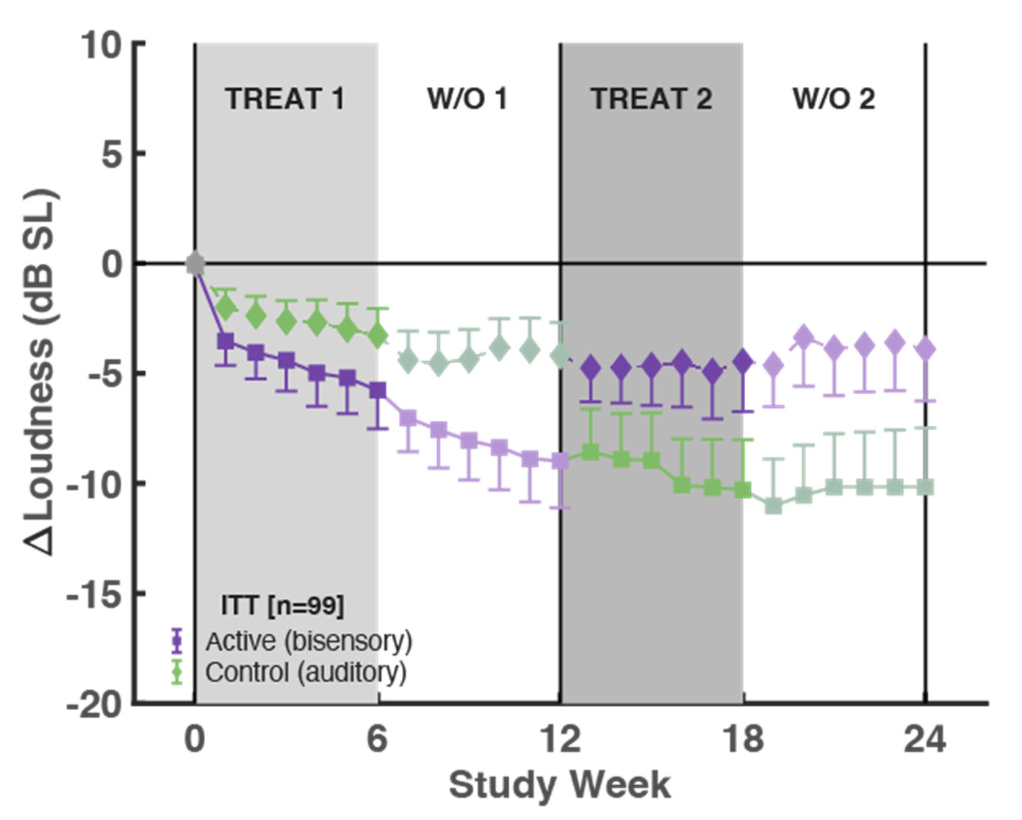
In numerous parts of the paper, there is reference to an average 14.1 dB loudness reduction for the Per Protocol (PP) group at week 12 for group 1 (e.g. in the "Results" section). But the graphs, e.g. Fig 3b, only show an 11 dB reduction. Do you know why this is the case?
This appears to be a mislabelling in the paper. The 14.1 value actually refers to the TFI, while the loudness should be -10.9, consistent with the text and figures.
According to the paper, "Given the large carry-over effect due to the longevity of the response, only period 1 was further analyzed per the standard procedure". The results from period 2 seems to be quite a bit different than period 1. I interpret the tinnitus loudness graph to mean that the group that experienced the treatment in weeks 1-6 did much better than the group that experienced the treatment in weeks 13-18. I don't see that there should be any particular reason for this, but I believe that the paper says it is due to the significant carry-over effect (?). The paper focuses its attention mostly on the results of the first group; can you elaborate on why this is the case?
As explained above in the question about a cross-over study, I’ll provide the same explanation again.
A crossover study is the strongest possible design for a clinical trial, but only if there is recovery (or return to baseline) after the first treatment period during the washout phase. This worked well for the first pilot study with 4 weeks of treatment (Marks et al, 2018). For that study, the majority of subjects’ tinnitus had mostly returned to baseline after the 4 week washout. For the second study we used 6 weeks of treatment and 6 weeks of washout before the crossover as we expected that we would see a return to baseline after 6 weeks of washout. We did not expect that the treatment would work so well after 6 weeks that there was no return to baseline during the washout. What we saw was that the treatment after 6 weeks was so long lasting that we did not see recovery. In fact, we saw a continued treatment effect up to 12 weeks (and beyond). For that reason the crossover did not work for the second study. This is called a “crossover effect” and it means that the second treatment period was biased by the first period. When that happens, it is not statistically valid to analyze the second half of the study. That is why we focused our analysis only on the first period.
What is a -6 dB tinnitus loudness reduction in real world terms? By how much does the tinnitus percept get reduced? Halved?
Yes, half as loud for every 6 dB decrease. So, -6 dB is perceived as 50% less loud, and -12 dB is perceived as 75% less loud.
Are there scientific metrics on how many participants (if any) reported their tinnitus as becoming lower in pitch or less harsh, even if not less loud?
Yes, subjects did show a lowering of the primary pitch of their tinnitus during the active treatment but not the sham treatment (see Marks et al, 2018).
Why was TFI reduction comparatively modest, wouldn't you think if people's tinnitus loudness reduced as much as in the study, the TFI reduction would be also much higher?
The TFI reduction was not modest, and was as much as 18 points lower than baseline TFI score at the 12 week time point. This is greater than the minimum value for clinical significance, and we saw that TFI reduction moved in parallel with reductions in tinnitus loudness.
How did the participants of the study react to their improvements? Particularly the two participants that went away without any symptoms, did they pre-order the device right away?
Yes, subjects have asked if they could buy the device even for high cost.
Study Follow-Up
Are you planning any follow-up studies?
There will be real-world studies once the device is marketed.
Personal Questions for Dr. Shore
Can you tell us more about yourself and your professional background?
I was born in Johannesburg, South Africa. My first degree was in speech pathology and audiology. I practised for a few years before going back to school to get a master’s degree and then Ph.D. in physiology, with the aim to study the brain. I spent many years studying the anatomy and physiology of the cochlear nucleus and other central structures. Major discoveries from my lab included documenting the structure and function of somatosensory innervation to the auditory system and how it changed after cochlear damage. These studies led to the study of tinnitus circuitry.
Do you feel we're far from a full cure that returns people back to full silence? Do you think this is the first step?
I don’t think anyone experiences true silence. The goal to to return to a state in which people can function well.
What is the extent of your involvement at Auricle Inc.? How much does teaching and lecturing impact and affect the work you do at Auricle Inc.?
I am a co-founder and the chief scientific officer of Auricle. The company was started to commercialize the treatment developed in my lab. The other co founders are Jonathan Pearson (the CEO), under whose expert guidance we are working towards an FDA approval, and David Martel, a former bioengineering student in my lab and co-inventor of the treatment.
Since I am now retired to Professor emerita status, I am no longer lecturing at the university. But I am continuing research activities with some of my colleagues at the University of Michigan. This does not impact my work at Auricle.
Do you have tinnitus? If yes, how did you get it and how long have you had it? Does it bother you? Have you tested your own device? If so, did it improve your tinnitus (temporarily or permanently)?
I have very mild tinnitus that is not bothersome. I served as a subject to help develop the TinnTester application in the lab but have not tried the device myself – this would really be a conflict of interest.
Do people over at Auricle Inc. ever go through Tinnitus Talk and use it as inspiration to help those in need?
I do not generally participate in social media forums.
Have you yet had the chance to take time to celebrate your breakthrough? Do you anticipate what impact your work will have?
I am too busy answering questions. 😄
Can we nominate you for the Nobel Prize?
My permission is not required.
Copyright © Tinnitus Hub Inc. All Rights Reserved.
Information published on this site is not intended as a substitute for medical advice. For medical care, consult a medical professional.